Origin of MCF-7 Cell Line
MCF-7 is a cell line that was first isolated in 1970 from the breast tissue of a 69-year old Caucasian woman. Of the two mastectomies she received, the first revealed that the removed tissue was benign. Five years later, a second operation revealed a malignant adenocarcinoma in a pleural effusion from which tissue was taken that would eventually result in the MCF-7 cell line. The donor was treated for breast cancer with radiotheraphy and hormonotherapy.
MCF-7, a widely studied epithelial cancer cell line derived from breast adenocarcinoma, has characteristics of differentiated mammary epithelium. MCF7 cells can be used for detecting PI3K and MAPK involvment, along with easy detection of ERK and Akt phsophorylation. Also, via cytoplasmic estrogen receptors, these cells have the ability to process estradiol.
Uses for MCF-7 Cell Line
MCF-7 cells are useful for in vitro breast cancer studies as a result of the cell line retaining several ideal characteristics particular to the mammary epithelium. These include the ability for MCF-7 cells to process estrogen in the form of estradiol via estrogen receptors in the cell cytoplasm. This results in the MCF-7 cell line being an estrogen receptor (ER) positive cell line. MCF-7 is also progesterone receptor positive and HER2 negative.
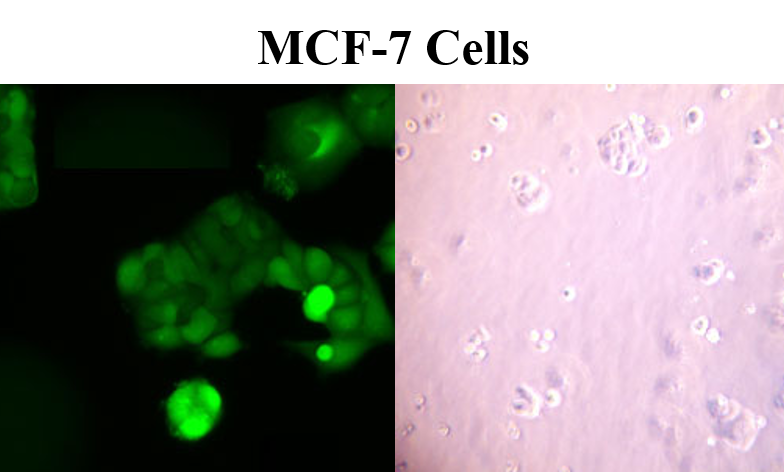
Characteristics of MCF-7
In addition to retaining estrogen sensitivity, MCF-7 cells are also sensitive to cytokeratin. They are unreceptive to desmin, endothelin, GAP and vimentin. When grown in vitro, the cell line is capable of forming domes and the epithelial like cells grow in monolayers. Growth can be inhibited by using tumor necrosis factor alpha (TNF alpha) and treatment of MCF-7 cancer cells with anti-estrogens can modulate insulin-like growth factor proteins, which ultimately have the effect of a reduction in cell growth. Scientists have found that although MCF-7 cells are easy to propagate, they are generally a slow-growing population. MCF-7 doubling time is typically 30-40 hours.
MCF-7 cells are fairly large adherent cells, with typical cell size measuring 20-25 microns. The recommended medium is EMEM containing 2 mM L-glutamine, 0.01 mg/mL bovine insulin (90%), fetal bovine serum (10%) and Earle’s BSS containing 1.5 g/L sodium bicarbonate, 0.1 mM non-essential amino acids and 1 mM sodium pyruvate.
Stability of cell line
Genetically, the MCF-7 line is not exactly the same as the intial isolated clone. Originally, it was described as having a karyotype containing 85 chromosomes which has since been reduced by 16 chromosomes.
Today’s MCF-7 cell line has a karyotype containing 69 chromosomes. Furthermore, there are genetic
discrepancies between the MCF-7 cell line from the Michigan Cancer Foundation and the ATCC cell line. This indicates that the ATCC cell line is of a different source than the other MCF-7 cell lines.
MCF-7 Xenograft Model
Validated breast cancer MCF7 xenograft model was established by Altogen Labs (Austin,TX) and commercially available, please see the following link for MCF-7 in vivo xenograft model laboratory testing service:
MCF-7 Xenograft Model
Transfection Protocol
An optimized MCF-7 Transfection Kit is available from Altogen Biosystems, which includes:
- MCF-7 Transfection Reagent (0.5 ml / 1.5 ml / 8.0 ml)
- Transfection Enhancer (0.5 ml), and
- Complex Condenser (0.5 ml)
This reagent is optimized for transfection of DNA and RNA into MCF-7 cells following either a standard or reverse protocol. The protocol for a 24-well plate to transfect MCF-7 cells is as follows:
- Plate 7,500-12,000 MCF-7 cells per well in 0.5 ml of complete growth medium 12-24 hours prior to transfection
- Wash with 1xPBS and add 0.5 ml of fresh growth medium
- Prepare transfection complexes by mixing 40 µl of serum-free medium, 5.5 µl of transfection reagent, and (referred to a final volume including growth medium)
- 750 ng DNA (or mRNA), or
- 30 nM – 50 nM of siRNA (or microRNA)
- Incubate transfection complexes at RT for 15-30 minutes
- Optional: Add 2 µl of Complex Condenser. This reagent increases transfection efficiency by reducing the size of transfection complex; however, it may increase cell toxicity
- Add prepared transfection complexes to 0.5 ml of complete growth medium with MCF-7 cells (from step 2)
- Incubate cells at 37ºC in a humidified CO2 incubator
- Assay for phenotype or target gene expression 48-72 hours after transfection
Optional: Adding Transfection Enhancer reagent can increase transfection efficiency. Add 2 µl of Transfection Enhancer reagent 12-24 hours after transfection
If the viability of MCF-7 cells being transfection is affected at 16-24 hours post-transfection, changing the growth medium and eliminating redundant exposure of cells to transfectant can decrease the level of cytotoxicity
MCF-7 Molecular Profile
MCF-7 breast cancer cells are estrogen E2-sensitive cells expressing high levels of ERα transcripts and very low levels of ERβ. MCF7 cells increase expression of estrogen receptor (ER) in the absence of estrogens. Response to E2 of MCF7 cells was demonstrated to be dependent on autocrine factor activating the insulin-like growth factor receptor (IGF-IR).
The growth of breast cancer cells is controlled by PR & ER as well as plasma membrane-associated growth factor receptors. Two particularly important members of this large family are EGFR activated by EGF, and the epidermal growth factor receptor 2 (HER-2).
Links
MCF-7 Xenograft CRO Services
Lab Techniques, Protocols, and Research Methods
MCF-7 cell line (Wikipedia)
Berkeley Lab MCF7 Breast Cancer Cell Line
DSMZ cell line data on MCF-7
Stable Cell Line Generation Services
Research
- Acquired Tamoxifen Resistance in MCF-7 Breast Cancer Cells Requires Hyperactivation of eIF4F-Mediated Translation. Fagan DH, Fettig LM, Avdulov S, Beckwith H, Peterson MS, Ho YY, Wang F, Polunovsky VA, Yee D. Horm Cancer. 2017 Jun 2.
- The impact of anticancer activity upon Beta vulgaris extract mediated biosynthesized silver nanoparticles (ag-NPs) against human breast (MCF-7), lung (A549) and pharynx (Hep-2) cancer cell lines. Venugopal K, Ahmad H, Manikandan E, Thanigai Arul K, Kavitha K, Moodley MK, Rajagopal K, Balabhaskar R, Bhaskar M. J Photochem Photobiol B. 2017 May 24;173:99- 107.
MCF-7 Cells Culture | MCF-7 Culture Protocol | MCF-7 Transfection Reagent